Quality
UMA Precision Machining is an ISO 13485:2016 Certified and preferred contract manufacturer of components for OEM manufacturers. UMA Precision Machining is committed to providing components and assemblies that meet the specified applicable requirements of our customers. We will maintain the effectiveness of the Quality Management System by prioritizing and executing quality objectives, organizational communication, and planned reviews for effectiveness. Examples as follows:
- Deploy lean manufacturing/Six Sigma tools
- Validated processes
- IQ, OQ, PQ procedures
- Process Failure Modes Effect Analysis (PFMEA)
- Product/process control plans
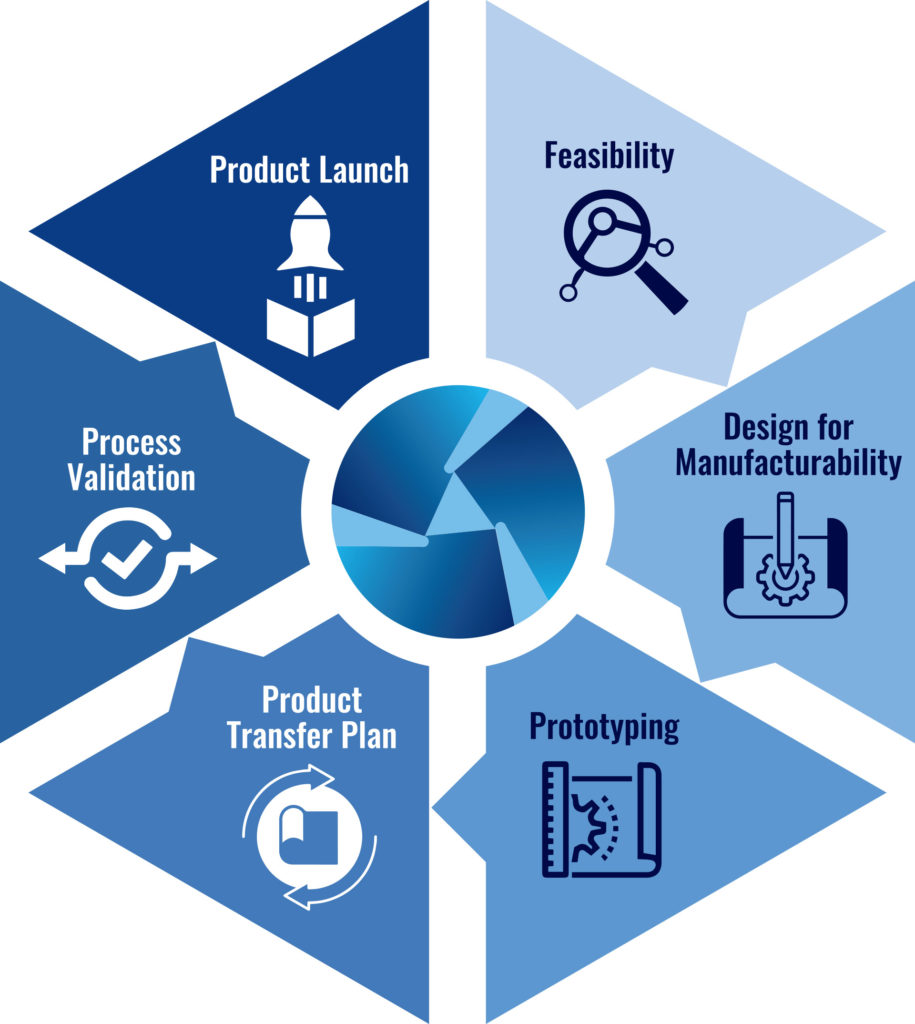
UMA Product Development Process

Teamwork
We partner with our customers on their product designs and specifications to offer creative, value-added solutions for design-for-manufacturing process development, assisting towards optimal results for prototypes, pre-production (including validation requirements) and production.

Assurance
Each operation of every job is under the watchful eye of UMA Precision Machining’s quality management team – assuring each operation meets or exceeds specifications. At UMA Precision Machining, quality control is more than a statistic…it is embedded in our work environment; it is our spirit; it is our culture.

Testing & Inspection
UMA Precision Machining has a full complement of Quality Inspection Equipment and Instruments – Calibrated to industry standards and comply with testing and inspection requirements.
